Explore our network of country and industry based websites to access localized information, product offerings, and business services across our group.
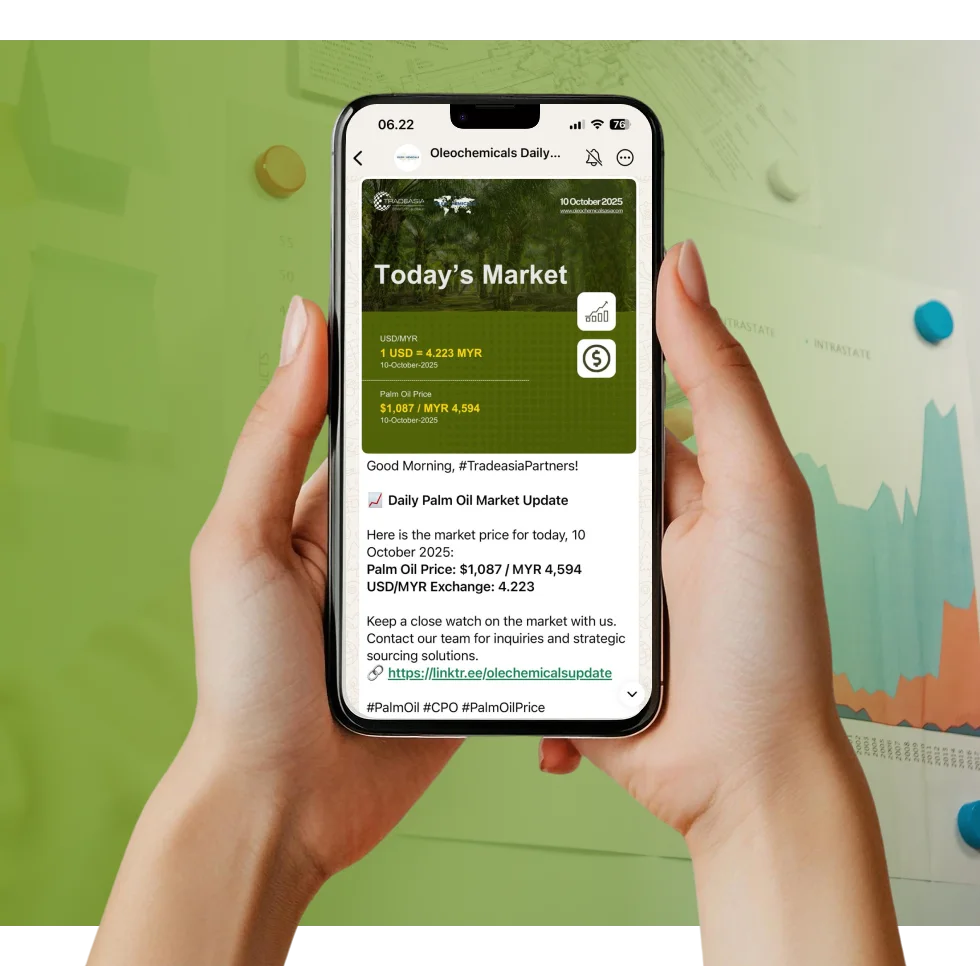
Access reliable chemical market information through our update channels.
Real-time Updates
Daily Updates
Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Log in now to access technical product documents from our product range.
Don't have an account? Sign Up Here
Home All Products Methyl Oleate
|
Origin |
: Indonesia |
|
IUPAC Name |
: Methyl (Z)-octadec-9-enoate |
|
Cas Number |
: 112-62-9 |
|
HS Code |
: 2916.15.90 |
|
Formula |
: C19H36O2 |
|
Appearance Name |
: Yellowish Oily Liquid |
|
Common Names |
: Oleic acid methyl ester |
|
Packaging |
: Drum, ISO, Bulk |
For more detailed information including pricing, customization, and shipping:
Brief Overview
Methyl oleate is a fatty acid methyl ester with a chemical formula of C19H36O2. The chemical compound is an oily liquid with a yellowish color and has a fatty odor. In addition, the compound is immiscible with water and miscible with chloroform, ether, and alcohol.
Manufacturing Process
The fabrication of methyl oleate is conducted by condensation of fatty acids with methanol. The fatty acid that contribute to the formation of methyl oleate is the oleic acid. By the addition of methanol, the chemical ester methyl oleate is formed. As by-products of fatty esters are formed, the compound is distilled and crystalized to obtain a purer concentration of methyl oleate.
Intermediate Chemicals
Methyl oleate is utilized as an intermediate chemical to fabricate detergents. In addition, the compound is used to create emulsifiers, wetting agents, plasticizers, and other industrial chemicals (rubbers and waxes).
