Explore our network of country and industry based websites to access localized information, product offerings, and business services across our group.
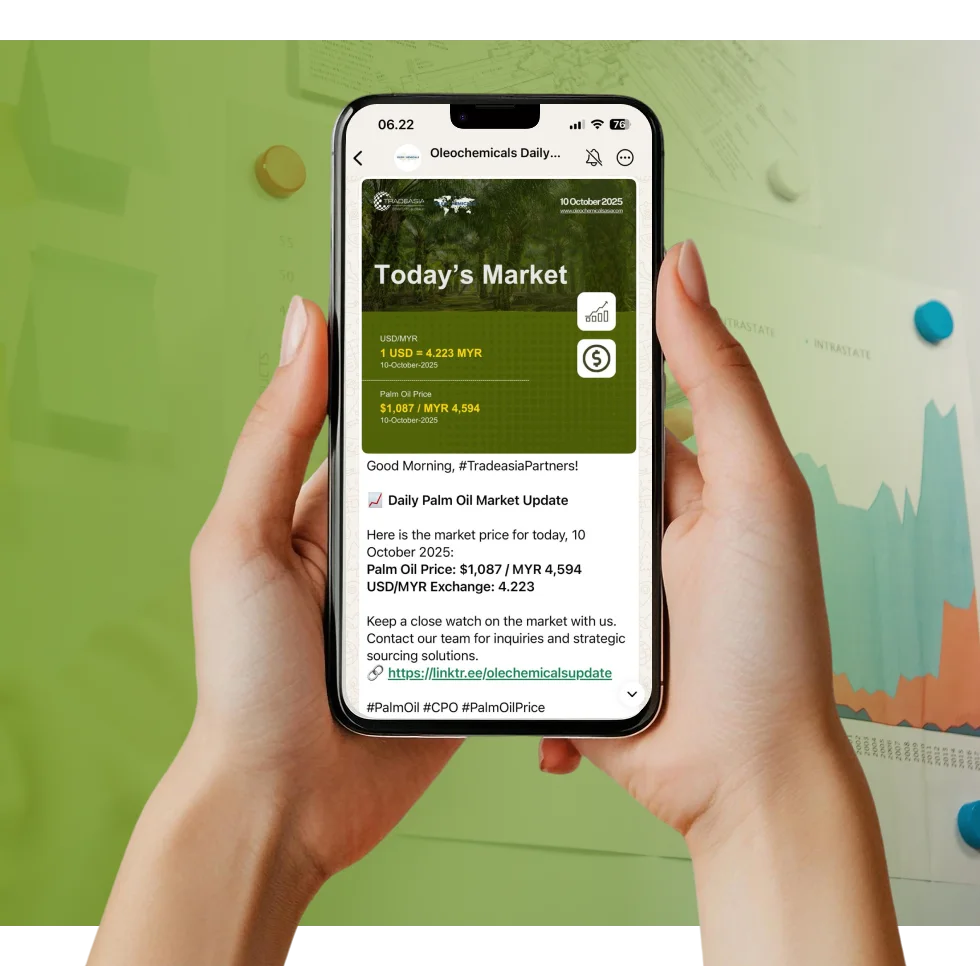
Access reliable chemical market information through our update channels.
Real-time Updates
Daily Updates
Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Log in now to access technical product documents from our product range.
Don't have an account? Sign Up Here
Home All Products Methyl Caprate
|
Origin |
: Indonesia |
|
IUPAC Name |
: Methyl decanoate |
|
Cas Number |
: 110-42-9 |
|
HS Code |
: 2915.90.90 |
|
Formula |
: C11H22O2 |
|
Appearance Name |
: Colorless, Clear, Oily Liquid |
|
Common Names |
: Capric acid methyl ester |
|
Packaging |
: PE, Drum, ISO, Bulk |
For more detailed information including pricing, customization, and shipping:
Brief Overview
Methyl caprate also known as methyl decanoate is a methyl acid that has a molecular formula of C11H22O2. At room temperature, methyl caprate is a colorless liquid oil with a melting point of -11 degrees centigrade and soluble in organic solutions such as ethanol and dimethyl formamide. The chemical compound is found naturally as a scent component in blue cheese and a flavor constituent in cassava.
Manufacturing Process
Methyl caprate is fabricated by the esterification process between triglyceride and methanol. An additional method is possible by isolating methyl caprate from the transesterification process between triglycerides/fatty acid and methanol. Initially extract of palm/vegetable oil undergoes decomposition into esters and glycerin. The various components of esters are purified by the distillation process. As glycerine is removed, the methyl caprate is isolated by separating the compound based on the boiling point. The methyl caprate distillate is collected from the distillation column.
Pharmaceutical Applications
Methyl caprate has a bactericidal property used in pharmaceutical products.
Chemical Intermediate
The compound is used as a chemical intermediate to manufacture capric acid. Capric acid as a compound is utilized in fabricating food flavoring products. Furthermore, methyl caprate is used as a chemical intermediate for manufacturing wetting agents, emulsifiers, stabilizers, and lubricants.
