Explore our network of country and industry based websites to access localized information, product offerings, and business services across our group.
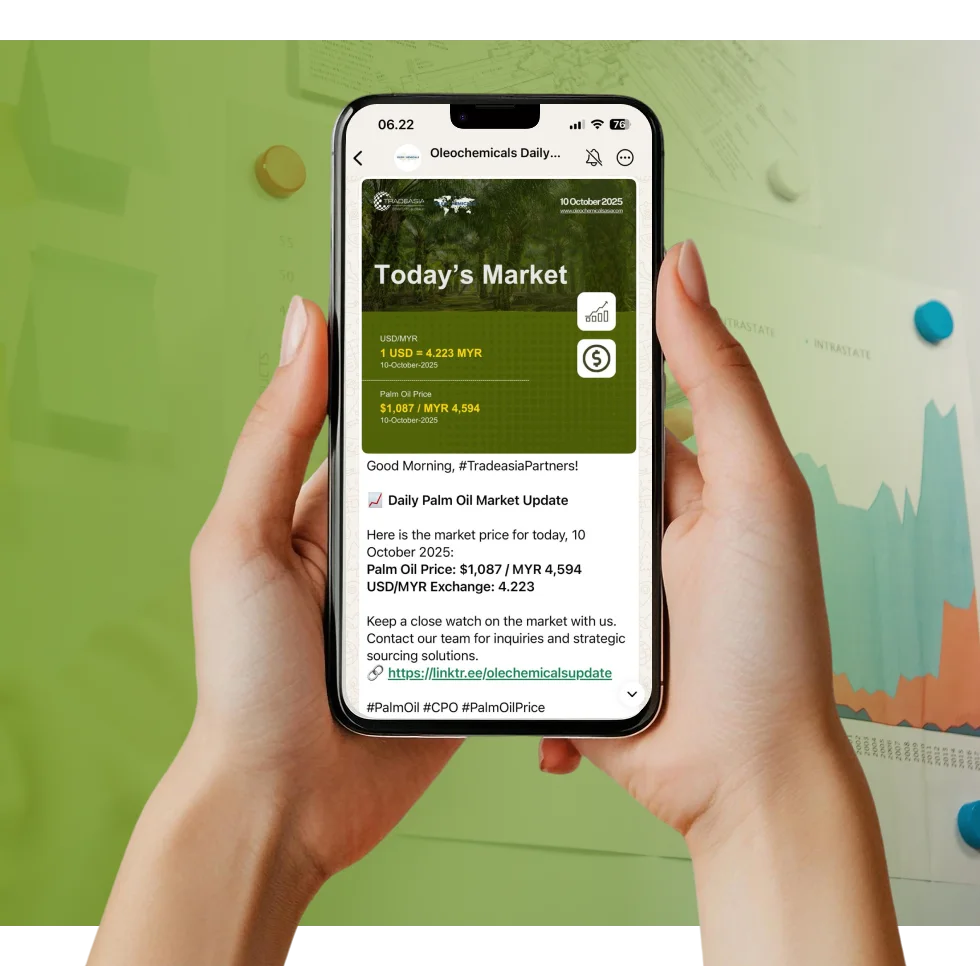
Access reliable chemical market information through our update channels.
Real-time Updates
Daily Updates
Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Log in now to access technical product documents from our product range.
Don't have an account? Sign Up Here
Home All Products Cetyl Palmitate
|
Origin |
: Indonesia |
|
IUPAC Name |
: Hexadecyl hexadecanoate |
|
Cas Number |
: 540-10-3 |
|
HS Code |
: 3404.90.90 |
|
Formula |
: C32H64O2 |
|
Appearance Name |
: White Crystalline Powder |
|
Common Names |
: Palmityl palmitate |
|
Packaging |
: 700 Kg Polypropylene Big Bags |
For more detailed information including pricing, customization, and shipping:
Brief Overview
Cetyl palmitate is known as an ester formulated from the reaction between carboxylic acid and alcohol. The compound is naturally found as a component of a the rare sperm whale wax called spermaceti. Cetyl palmitate appeared as a white crystalline powder that easily dissolves in hot acetone.
Manufacturing Process
Cetyl palmitate is manufactured by mixing hexadecenoic acid and hexadecanol with 1:1 molar ratio. The mixture is superheated into a molten state and mixed in a uniform motion. The mixture is then cooled to room temperature and heated once again to 350 degrees centigrade. The heat is maintained until 30 minutes to yield cetyl palmitate as the product.
Cosmetic Applications
Cetyl palmitate has the properties to bind water on the surface of the skin. The layer of lipids covering the skin helps to retain the moisture of the skin; therefore, the chemical compound functions as an emollient. The compound’s water retaining possible is able to maintain the texture and creaminess of cosmetic products. On the other hand, cetyl palmitate is a good thickener. The thickening properties of the compound allows to produce viscous emulsion by adding the desired texture to the mixture.
