Explore our network of country and industry based websites to access localized information, product offerings, and business services across our group.
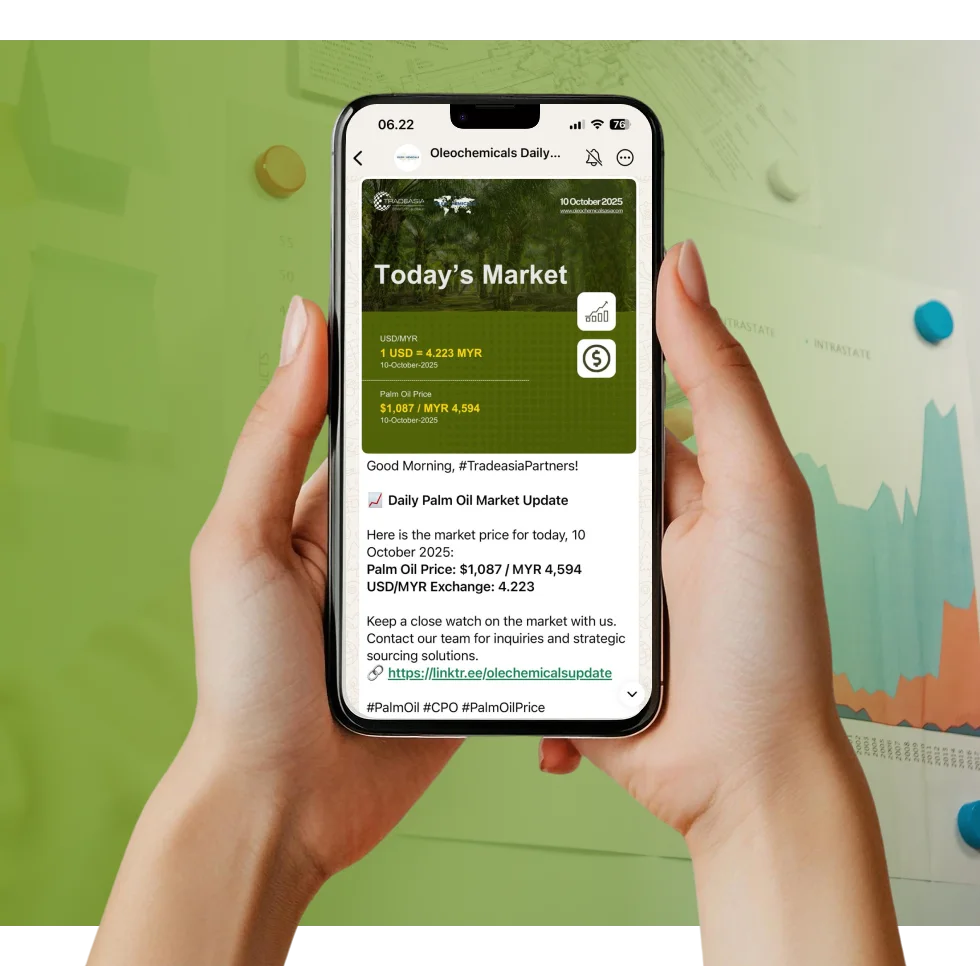
Access reliable chemical market information through our update channels.
Real-time Updates
Daily Updates
Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Log in now to access technical product documents from our product range.
Don't have an account? Sign Up Here
Home All Products Refined Glycerine 99.5% Min Mixed Animal Fat
|
Origin |
: Brazil |
|
IUPAC Name |
: Propane-1,2,3-triol |
|
Cas Number |
: 56-81-5 |
|
HS Code |
: 2905.45.00 |
|
Formula |
: C3H8O3 |
|
Appearance Name |
: Clear Colorless Liquid |
|
Common Names |
: 1,2,3-propanetriol, Glycerol |
|
Packaging |
: 20 MT / Flexi Bag |
For more detailed information including pricing, customization, and shipping:
BRIEF OVERVIEW
Refined Glycerine, also known as glycerol or glycerin, is a straightforward sugar alcohol with three hydroxyl groups, resulting in its water solubility and hygroscopic nature. This clear, colorless, and odorless liquid, with a high boiling point and viscosity, is naturally sweet and low in toxicity. Boasting a density of 1.26 g/cm³, it is derived from petrochemical feedstock and finds applications in various industries, including food, medicine, cosmetics, personal care, and biodiesel production.
MANUFACTURING PROCESS
Recovery of fat
Animal fats are collected from slaughterhouses. The fat tissue from animal parts is cooked. The elevated temperature ensures the fat to be released in liquid state and promotes cell rupture. On the other hand, an alternative method requires partial heating and the mechanical rupture of the fat tissue to release fat.
Degumming
The degumming process of animals is the removal of dissolved phosphatides through centrifugation. Water is added to the oil that allows the precipitation of phosphatides. The newly formed precipitate is heavier as phosphatides readily absorb water. The centrifugation process separates the heavy phosphatides from the oil phase to the water phase.
Bleaching
Bleaching of oil is required to release unwanted pigment and other oxidizing agents that may alter the appearance of the product. Bleaching is performed by adding charcoal or earth clay. The existing pigment from the oil will be absorbed by the charcoal. On the other hand, an additional method by chemical reaction is required for edible oils (oxidation and reduction reaction).
Deodorization
The adsorbed oils are further treated for scent and foul odor removal. Steam distillation is utilized to remove any volatile impurities that contribute to the unpleasant smell. Steam distillation makes use of the nonvolatile animal fat to remove any aldehydes, ketones, and other compounds that are classified as an impurity. In a distillation chamber, high pressure is injected under vacuum conditions around 180-250 degrees centigrade. The non-volatile triglyceride remains unchanged while volatile impurities cascade upwards and are removed from the animal fat.
Transesterification
The animal fat is decomposed by hydrolysis reaction under high temperature and pressure. The process utilizes water to break down the chains of triglycerides into glycerol/glycerine and chains of fatty acid. In this step, glycerine becomes available for extraction; however, further refining is required for the newly made glycerine to be called refined glycerine.
Separation
The mixture of glycerine and a chain of fatty acids is further refined by a series of separation processes. The oil mixture is distilled to separate the fatty acids from the desired glycerine. The distillation column is heated to promote the evaporation of both glycerol and fatty acid. Glycerine has lower boiling point; therefore, glycerine will evaporate readily at a certain range temperature whereas the chains of fatty acid will remain as liquid.
Pharmaceuticals Industry
Glycerine serves as a crucial raw material in pharmaceuticals, contributing to medications, suppositories, cough remedies, and anesthetics.
Cosmetic Industry
In cosmetics, glycerine acts as a water retainer, preserving product softness, creaminess, and storage life.
Food Industry
Glycerine is utilized in athletics for hydration and serves as a sweetener in various food products.
Industrial Applications
Glycerine is researched for fuel additives, decreasing harmful emissions. Its lubricating properties enhance machine performance, reduce friction, dissipate heat, and prevent machinery overheating. Glycerine's economic viability makes it valuable in chemical intermediates and industrial applications.
